On Thursday, August 16, the National Institute of Allergy and Infectious Diseases (NIAID) announced that vaccinations have begun in a first-in-human trial of an experimental live, attenuated Zika virus vaccine.
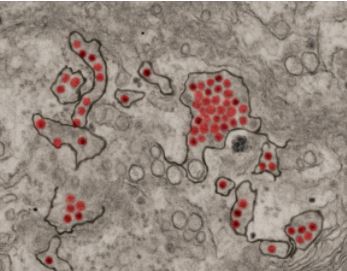
Zika virus particles (red) shown in African green monkey kidney cells. (Image courtesy of NIAID)
On Thursday, August 16, the National Institute of Allergy and Infectious Diseases (NIAID) announced that vaccinations have begun in a first-in-human trial of an experimental live, attenuated Zika virus vaccine. The trial will enroll a total of 28 healthy, non-pregnant adults ages 18 to 50 at the Johns Hopkins Bloomberg School of Public Health Center for Immunization Research in Baltimore, Md., and at the Vaccine Testing Center at the Larner College of Medicine at the University of Vermont in Burlington. NIAID is sponsoring the trail.
Read more on
National Institutes of Health,
United Press International Online.